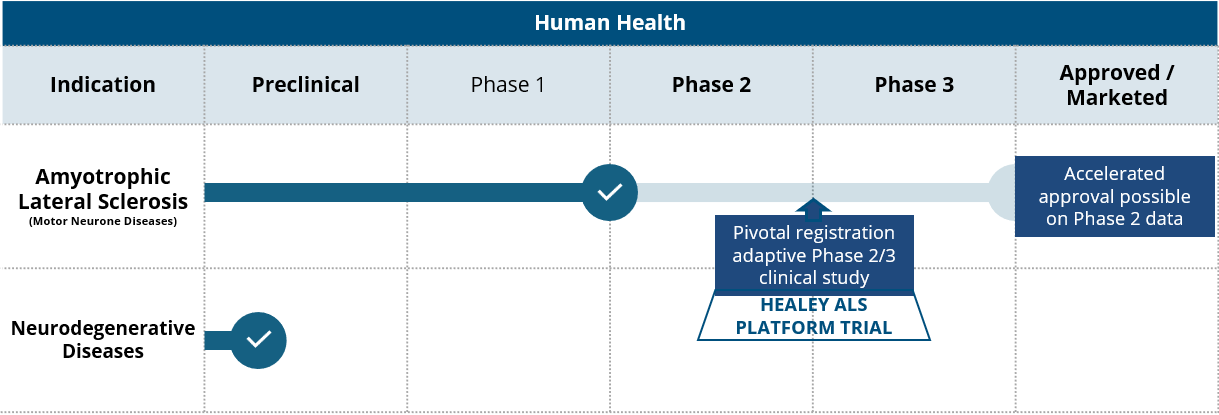
Product Pipeline
In mammalian cells, monepantel is a potent inhibitor of the mTOR pathway which plays a central role in cell growth and proliferation of cells. In degenerating neurons the mTOR pathway regulates autophagic flux and the recycling of cellular macromolecules.
PharmAust’s Therapeutic Pipeline
Monepantel is an approved veterinary product being repurposed for the treatment of motor neurone disease in humans. It also has potential in other neurodegenerative diseases, such as Alzheimer’s and Parkinson’s disease.